
In patients with concurrent celiac disease or IBD, the diagnosis of microscopicĬolitis is often difficult. Under the umbrella of microscopic colitis have anĪssociation with other autoimmune disorders like celiac disease, rheumatoid arthritis, thyroid disease or diabetes mellitus. Of lymphocytic colitis is infiltration of the colonicĮpithelium with lymphocytes. 
It is easy to distinguish collagenous colitis from lymphocytic colitis by the presence of a thick subepithelial collagen band whereas the telltale sign 1 Microscopic colitis can be confused with diseases that present similarly, such as celiac disease, IBS or IBD.Ī colonoscopy is often normal and biopsies of theĬolonic mucosa are required to make a definitiveĭiagnosis. It affects women more commonly than men and presents usually above the age of 65. The names describe the change to the colonic mucosa that is not due to an infectious cause, ultimately causing chronic watery, non-bloody diarrhea without other physical findings. The term microscopic colitis encompasses two separate diagnoses: lymphocytic colitis and collagenous colitis. In this paper we will discuss current methods of diagnosis, confounding issues, and treatment. Treatment is usually aimed at shortening the duration of symptoms. Its diagnosis can be complicated further with concurrent celiac disease or IBD. (8) Anemia can be a presenting and/or significant.The vague presentation of microscopic colitis is similar to celiac disease, irritable bowel syndrome (IBS), or inflammatory bowel disease (IBD). Adult celiac disease can be a cause of malabsorption of several nutrients in addition to having a malignant potential. (1-6) First described in 1888 by Samuel Gee, (7) adult celiac disease has now been well recognized as a disease characterized by damage to the small bowel mucosa induced by gluten. The different lesions responsible for chronic blood loss include both upper and lower gastrointestinal tract sources with causes and incidences varying widely among different studies. Occult, chronic blood loss from the gastrointestinal tract is the most common cause of iron-deficiency anemia. Given the treatable nature of celiac disease, it should be screened for in patients with unexplained iron-deficiency anemia with or without hemoccult-positive stools. A significant number of other gastrointestinal lesions amenable to therapy were also found on upper and lower endoscopy in these patients. Multiple findings were found in 32.3% of patients, and there were no findings in 28.5% of patients.Ĭonclusion: The prevalence of occult celiac disease in this prospective study of patients presenting with iron-deficiency anemia was 2.8%. Colonoscopic findings included colon polyps in 21.9%, diverticula in 10.4%, and hemorrhoids in 16.1%. Upper endoscopic findings included gastritis in 22.8%, gastric ulcers in 9.5%, duodenitis in 8.5%, esophagitis in 7.6%, Barrett's ulcer in 2.8%, duodenal ulcer in 2.8%, gastric polyp in 2.8%, and celiac disease in 2.8%. Forty-three and eighttenths percent of the men and 37.5% of women had hemoccult-positive stools, accounting for a total of 40.9% of the study patients. Nine patients were of mixed or unknown ethnic background. The demographics of this study population included 36 blacks, 38 Hispanics, and 22 whites. Fifty-seven men (mean age, 51.6 yr) and 48 women (mean age, 54.1 yr) constituted the study population. Results: One hundred five of 139 consecutive patients with iron-deficiency anemia met the inclusion criteria and were enrolled in the study. A serum immunoglobulin A antiendomysial antibody test was to be performed in those patients with a positive small bowel biopsy to confirm the diagnosis of celiac disease. All patients underwent upper endoscopy with more than two biopsies of the distal duodenum and colonoscopy. Patients were excluded for documented prior erosive, ulcerative, or malignant disease of the gastrointestinal tract, previous gastrointestinal surgery, overt gastrointestinal bleeding within the past 3 months, or inability to access the duodenum for biopsy. Inclusion criteria included iron-deficiency anemia as defined by a serum ferritin <25 ng/ml and anemia with hemoglobin <12 g/dl. Methods: Esophagogastroduodenoscopy with small bowel biopsies and colonoscopy were performed in all iron-deficiency anemia patients (including those with hemoccult-positive stools) referred to the gastroenterology service during a 2-year period (1998-2000). To more accurately define the true prevalence of this disorder in patients presenting with iron-deficiency anemia (with or without stool hemoccult positivity), we initiated this prospective study. 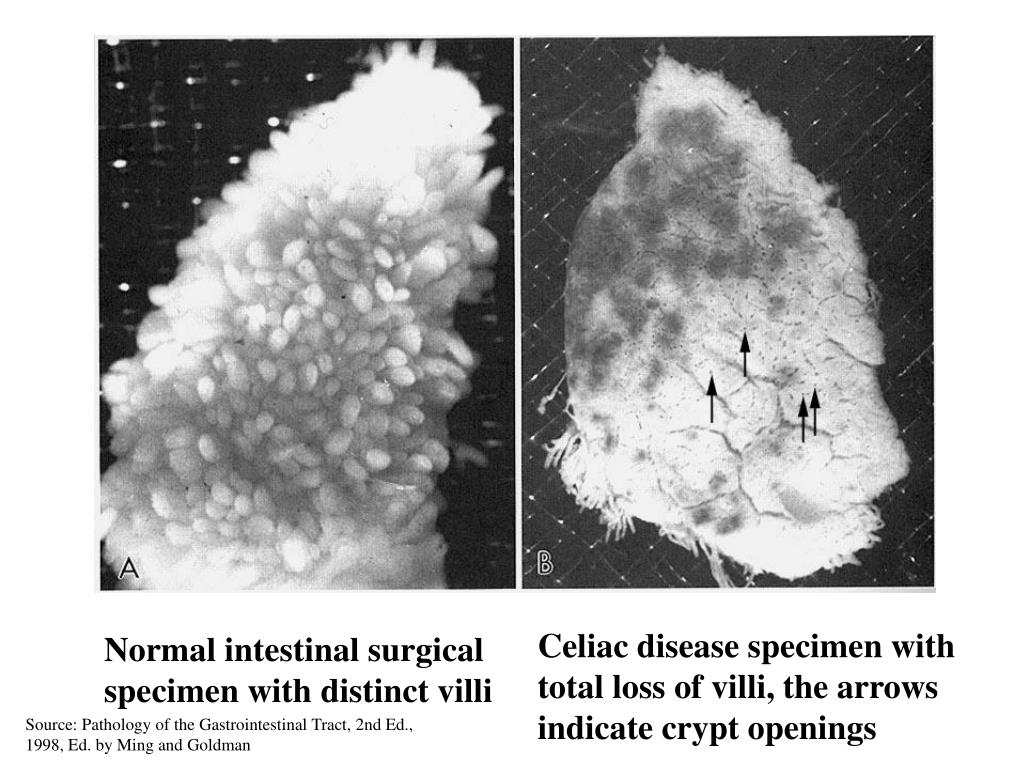
Most prior studies have been retrospective or screened only a selected population of patients with small bowel biopsies. Background: Occult celiac disease has been reported in 0 to 6% of adults presenting with iron-deficiency anemia.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
